
Medical 3-D Printing Update in Asia
Increasing demand for custom surgical implants and growing investment in R&D will help the sector experience significant growth over the next five years.

Increasing demand for custom surgical implants and growing investment in R&D will help the sector experience significant growth over the next five years.

Across Asia, government health and safety authorities have tightened regulations on medical devices this year, as markets continue to experience rapid change, and as the devices themselves gain sophistication and complexity at a blistering pace.

In general, the Asian markets have controlled the COVID-19 virus successfully outside of China, but its effect has still led to new developments and trends.

Across the continent, governments and hospitals are purchasing robotics systems from abroad as well as developing the technologies themselves.

The future of eye care will sit at the intersection of the digital and physical worlds.
With the exception of a few countries, Asia’s disease management industry is relatively unstructured.

While Western countries still have more medical device startups than the rest of the world, more new medtech startups are popping up in Asia than ever before.
As standalone software becomes increasingly sophisticated, a number of regulatory bodies have begun to draft separate guidelines to ensure quality control, expedite product approval, and improve patient access to new technologies.
As standalone software becomes increasingly sophisticated, a number of regulatory bodies have begun to draft separate guidelines to ensure quality control, expedite product approval, and improve patient access to new technologies.
Growth of the Asian cloud computing market is expected to accelerate as healthcare providers streamline their operations in today’s paperless world.
Growth of the Asian cloud computing market is expected to accelerate as healthcare providers streamline their operations in today’s paperless world.
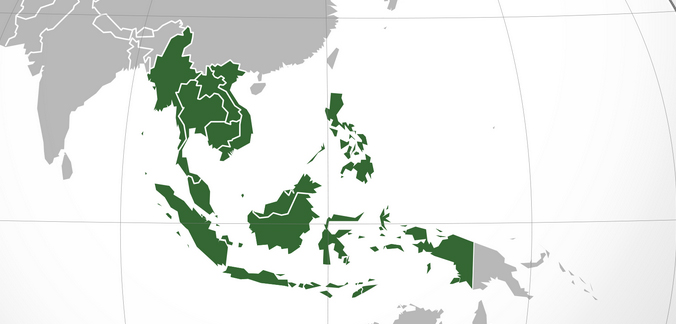
Foreign device companies can expect a big opportunity for expansion in Southeast Asia.