
Cybersecurity, COVID-19 and EU MDR Hot Topics of the 2020 MedTech Summer
Although the full impact of COVID-19 is uncertain, one sure thing is that industry continues to crave information to help them navigate EU MDR.

Although the full impact of COVID-19 is uncertain, one sure thing is that industry continues to crave information to help them navigate EU MDR.

The medical device industry is pressured to aid those stricken by the pandemic, while at the same time working to mitigate increased risks usually associated with hurried manufacturing and quality control procedures.

Being prepared significantly improves the odds that an organization will successfully manage and mitigate risk, sustain trust and loyalty, and protect and preserve their brand.

Recalls for failure are chronic in the medical device industry. The sources of those failures could originate anywhere in the supply chain. Fixing supply chain issues is ultimately the responsibility of the instrument’s maker.

The hot issues are related to compliance, EU MDR and recalls.

The number of recalls also increased 21.5% this quarter, according to Stericycle’s Q3 2019 Recall Index.
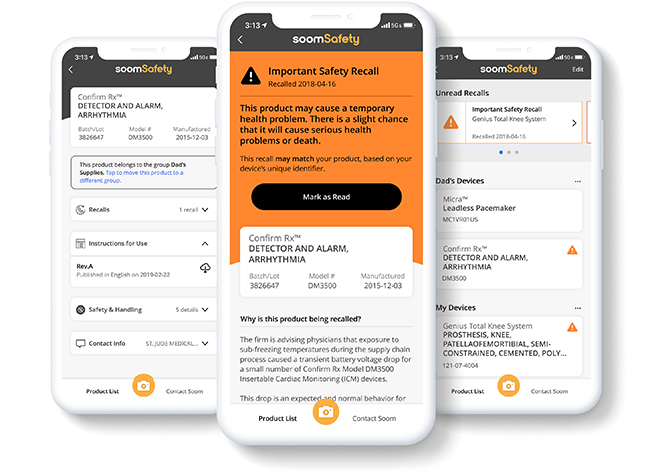
By simply scanning a barcode, SoomSafety gives users access to important product instructions, safety and recall information.

A recent project conducted by Northeastern graduate students looked at device recalls over the past five years and some of the common threads.

Medical device recalls were at their lowest quarter since Q4 2011, according to Stericycle’s recall index for Q4 2017.

When a software patch or device update cannot remedy a cyber threat, sometimes a recall is necessary.