
Balancing Compliance and Quality in Medical Device Manufacturing
As the pace of innovation further accelerates in 2021, and the need to get new devices to market intensifies, maintaining adherence to regulatory controls is not enough.

As the pace of innovation further accelerates in 2021, and the need to get new devices to market intensifies, maintaining adherence to regulatory controls is not enough.

The greatest challenge faced is an understanding of the intent of the written regulations and applying them correctly.

Participate in our new survey and have a chance to win complimentary access to the Computer Modeling & Simulation Virtual Conference on January 27–28, 2021.
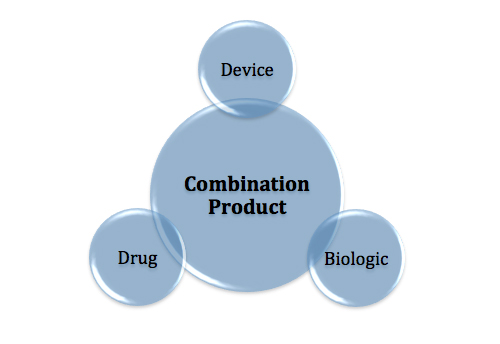
Understanding the GMP requirements of each component is critical to product and compliance success.

The technology behind remote cardiac device monitoring is becoming more convenient, responsive and connected across the digital ecosystem—including a new smartphone app.
No longer just “the big company advantage,” CM&S is an accepted and viable device industry path to market for large and small medical device makers alike.

Moving too quickly in the product development process can lead to mistakes.

No longer just “the big company advantage,” Arlen Ward discusses how CM&S is an accepted and viable device industry path to market for large and small medical device makers alike.

The center intends to facilitate partnerships, exchange knowledge and promote innovative regulatory approaches.

A Q&A with MedCrypt’s Axel Wirth sheds light on urgent problems that the medtech industry is facing regarding device security, but assures us that the sky is not falling.