
Navigating Global Regulations for SaMD
Decoding the legal framework for faster time-to-market.

Decoding the legal framework for faster time-to-market.

Hägen, who was a contributor to Medtech Intelligence, spent 28 years building BlackHägen Design to support clients in the development of “mission-critical” products. He was pivotal in the advocacy of human factors and good design practices for medical devices and was at the forefront of patient safety initiatives, helping to found the Industrial Designers Society of America (IDSA) Patient Safety Committee.

Reusable medical devices have many notable advantages. However, they’re not appropriate in all cases. This article discusses some factors designers should consider to make their products as profitable and useful as possible.

This article reviews real-world examples of product failures, presents reasons for these failures and provides potential solutions better governance, collaboration, and data gathering throughout the product lifecycle.

Today’s health monitoring and management systems rely on wires and batteries, and often are not continuously connected to an alert and communication system for patients and doctors. Wireless power will empower medical device manufacturers to develop sophisticated, smart IoT systems that will improve patient care and patient lives.
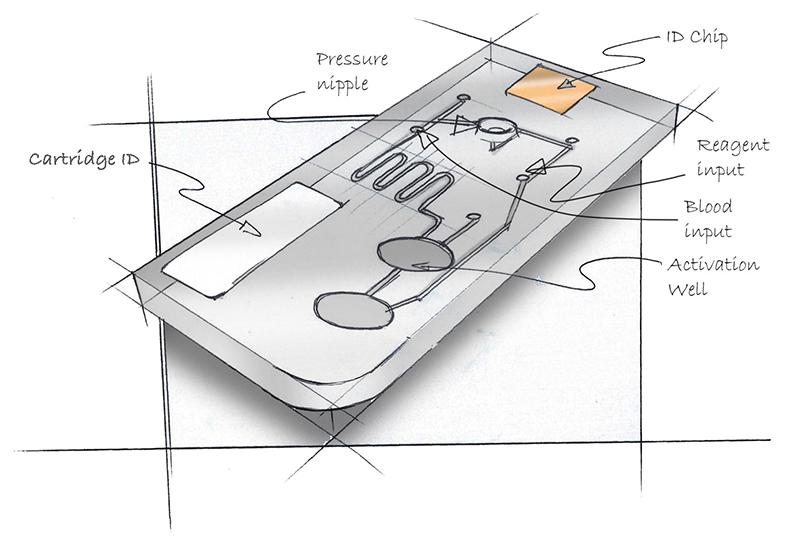
By examining the larger user experience involved in the microfluidics process, we can identify issues, and design these experiences to reduce the potential for user error, improve outcomes and create a simpler, more accessible process.
Adhesive selection can make or break the success of a stick-to-skin wearable device. This article reviews six questions to ask the next time you design a device intended for wear on fragile skin.

When a company does not take action to become more environmentally responsible, it risks losing access to global markets.

The adoption of this manufacturing process in healthcare saw a tremendous uptick in 2020.

Participate in our new survey and have a chance to win complimentary access to the Computer Modeling & Simulation Virtual Conference on January 27–28, 2021.