
ECRI Warns 70% of KN95 Masks from China Don’t Meet Standards, Puts Healthcare Workers at Risk
The non-profit organization states that 60–70% of KN95 masks tested do not filter 95% of aerosol particulates.

The non-profit organization states that 60–70% of KN95 masks tested do not filter 95% of aerosol particulates.

Across Asia, government health and safety authorities have tightened regulations on medical devices this year, as markets continue to experience rapid change, and as the devices themselves gain sophistication and complexity at a blistering pace.

Part one of this series looks at the claims brought against manufacturers of diagnostic test kits.

The medical device industry is pressured to aid those stricken by the pandemic, while at the same time working to mitigate increased risks usually associated with hurried manufacturing and quality control procedures.

User needs and expectations continue to advance. Medical device technology needs to keep pace. For a successful design, take the following requirements into consideration.

Investments in new processes and systems must satisfy the needs of the authorities and ensure patient safety and public confidence. Getting it right is likely to be expensive. but regulatory compliance shouldn’t be viewed purely as a cost center.

As the proliferation of connected and complex medical devices grows, healthcare providers are more susceptible to cyberattacks.

Simplifying the PMS process globally from end to end, can help medtech companies realize significant improvements across the board while keeping operating costs under control.
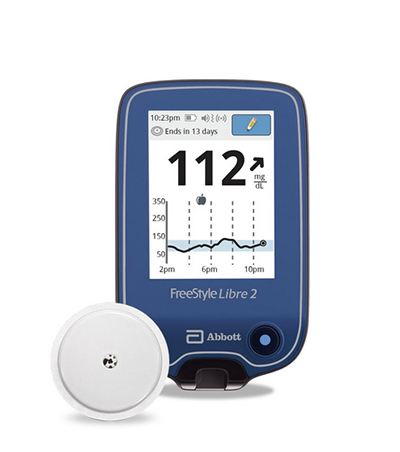
The FreeStyle Libre 2 now has indications for children ages four and older.

Medtech companies will need to collaborate more closely with other parties in healthcare, as the aftermath of the coronavirus pandemic will have a comprehensive reach throughout the entire ecosystem. MTI speaks with Johnson & Johnson VP Raymond Fryrear, M.D. on the topic.