
AI Applications
Artificial intelligence has numerous practical applications in diagnostic imaging; the key to making them work for clinicians and patients lies in developing and embracing integrated workflow networks.

Artificial intelligence has numerous practical applications in diagnostic imaging; the key to making them work for clinicians and patients lies in developing and embracing integrated workflow networks.

Peter O’Blenis, CEO of Evidence Partners, discusses the growing role—and challenges –of literature reviews in the medtech market.

Remote care in the home relies both on the quality of patient monitoring and on the insights provided to the care team. There is a real danger that data overload and alert fatigue will undermine otherwise well-designed remote patient monitoring (RPM) and Hospital at Home programs. The software platform and algorithms tasked with integrating and evaluating data must identify the data that matters, when it matters.
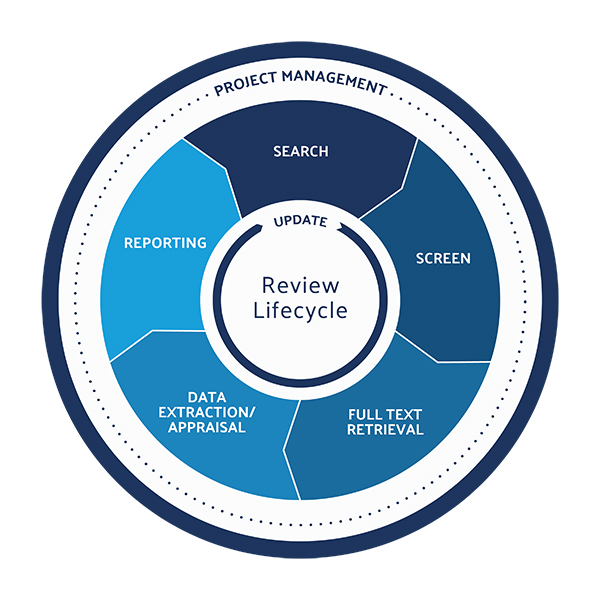
Double-digit growth of the company’s literature review software platform, DistillerSR, spurred $20 million in funding from the growth investment consortium led by Thomvest Ventures.

Next-generation, predictive analytic patient monitoring lowers healthcare costs, improves clinical outcomes and enhances the patient experience in hospital-at-home, post-acute care and chronic care management.

Randomized clinical trials are the gold standard for conducting clinical research. They are, however, fraught with issues and come with burdens to the patient, site and sponsor. Decentralized trials (DCTs) allow for a novel, agile and more cost-effective approach to an industry standard and traditional methodology. Here’s how to determine if a full or hybrid DCT is a good option for your clinical trial.

The COVID-19 pandemic exposed the necessity of virtual care and revealed its possibility as a more efficient means of administering treatment in an overwhelmed and understaffed infrastructure. Its fast-paced adoption highlighted the need for global standards and third-party certifications.

Embracing compliance is a continuous process, and investing in agile technologies that streamline workflows—especially in meeting EU MDR and IVDR requirements—is essential, says Lana Feng, Ph.D., CEO-founder of Huma.AI, a pioneer in a human-centered AI.
Diagnostic advances bode well for the imminent expansion of the global COVID-19 detection kits market, which is expected to be worth $8 billion by 2027.

Healthcare services are expanding their ability to leverage data for use cases such as diagnostics, personalized treatment, imaging analysis, patient trend analysis, outcomes predictions, automation and more.