Steady Improvements to Medical Device Quality in China
How CFDA enforces new regulations is critical to the future.
How CFDA enforces new regulations is critical to the future.
How CFDA enforces new regulations is critical to the future.
How CFDA enforces new regulations is critical to the future.
The increase in communicable diseases is elevating competition in the device arena.
Targeting this region could expedite your company’s growth.
Targeting this region could expedite your company’s growth.
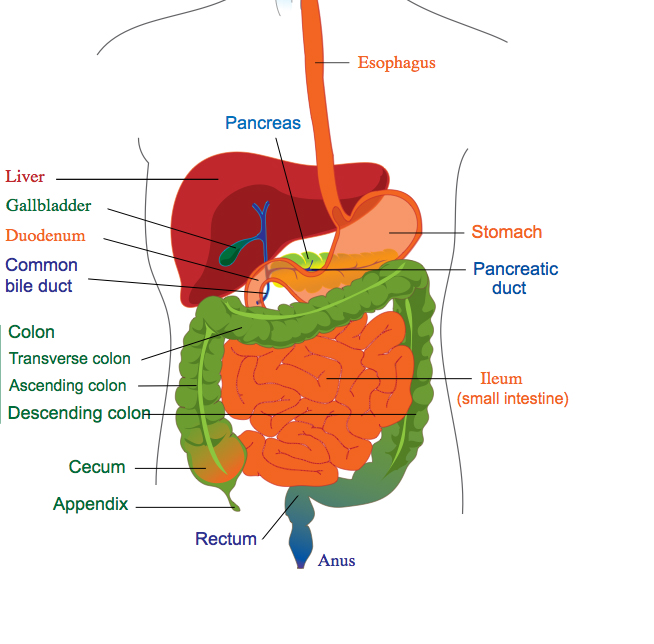
As the incidence of GI conditions grows, device companies in this segment should explore expansion in the region.
Companies will soon be able to capitalize on the government’s push to encourage investment in private healthcare facilities.
Companies will soon be able to capitalize on the government’s push to encourage investment in private healthcare facilities.

The booming industry is creating new opportunities for medtech manufacturers.

The rise in healthcare spending, jump in Western diseases, and increasing aging population are driving the market.
Achieving success in India involves patience and a firm grasp on the culture and regulatory environment.
Achieving success in India involves patience and a firm grasp on the culture and regulatory environment.
As the incidence of diabetes across Asia expands, demand for medical devices and drugs to treat the disease is intensifying.
As the incidence of diabetes across Asia expands, demand for medical devices and drugs to treat the disease is intensifying.
Remember, several basic regulatory approvals are required.
Remember, several basic regulatory approvals are required.