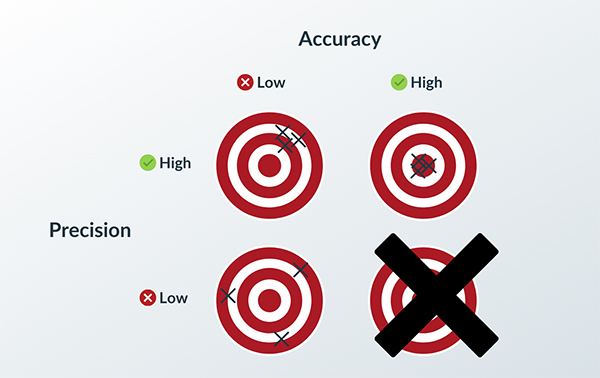
How to Achieve Quality through Process Validation
The key is to keep quality high and risk low.

The key is to keep quality high and risk low.

The pandemic has ushered in a novel era of cardiac health management, with remote patient monitoring at its forefront.

A review of common risks and pitfalls of incorporating artificial intelligence in medical devices and an overview of the regulatory framework.
Robust trends are expected to power the global digital diabetes management market.

During the COVID-19 crisis, digital forces like artificial intelligence have assisted the healthcare industry to focus on patient care while achieving improved efficiency during treatments.

Devices by themselves don’t improve outcomes. Better lifestyle integration is key to driving changes in patient compliance—embedding sensors into the sorts of devices people can use every day to increase opportunities for passive biometric capture and to facilitate therapeutics.
A global need for less invasive medical procedures and high efficacy treatments has outlined the developments of cardiovascular devices.

Beyond simply allowing for broader yet more efficient searches, leveraging software to automate literature reviews can organize references, assign screeners, and review screening decisions. This saves time, reduces bottlenecks, and, most importantly, leads to a highly transparent, standardized, and repeatable process that supports continuous CER and PER submissions across a product portfolio and for the life of a device.

In this series we examine the future of the medical device industry—from manufacturing to the consumer-patient experience.

Seamless and personalized diabetes care has become a new normal.