

FDA also supports the removal of the HeartWare HVAD System from the market.

FDA also supports the removal of the HeartWare HVAD System from the market.

The medtech regulatory environment in Europe has entered a new era. A Europe-wide medical device regulation has come into effect, presenting both challenges and opportunities for stakeholders in the sector.

As the pace of innovation further accelerates in 2021, and the need to get new devices to market intensifies, maintaining adherence to regulatory controls is not enough.
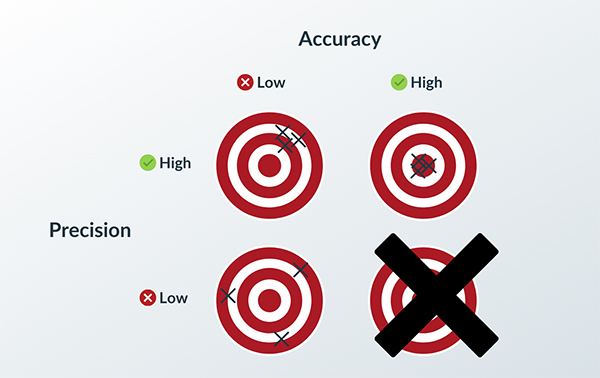
The key is to keep quality high and risk low.

A review of common risks and pitfalls of incorporating artificial intelligence in medical devices and an overview of the regulatory framework.
Robust trends are expected to power the global digital diabetes management market.

Digital health tools and technology have the potential to dramatically improve the patient care experience, but the shift requires purposeful coordination and planned architecture.

During the COVID-19 crisis, digital forces like artificial intelligence have assisted the healthcare industry to focus on patient care while achieving improved efficiency during treatments.

When a company does not take action to become more environmentally responsible, it risks losing access to global markets.

Devices by themselves don’t improve outcomes. Better lifestyle integration is key to driving changes in patient compliance—embedding sensors into the sorts of devices people can use every day to increase opportunities for passive biometric capture and to facilitate therapeutics.